Click here to buy a book photographic periodic table poster card deck or 3d print based on the images you see here.
Lattice parameter of pure iron at room temperature.
For elements that are solid at standard temperature and pressure the table gives the crystalline structure of the most thermodynamically stable form s in those conditions.
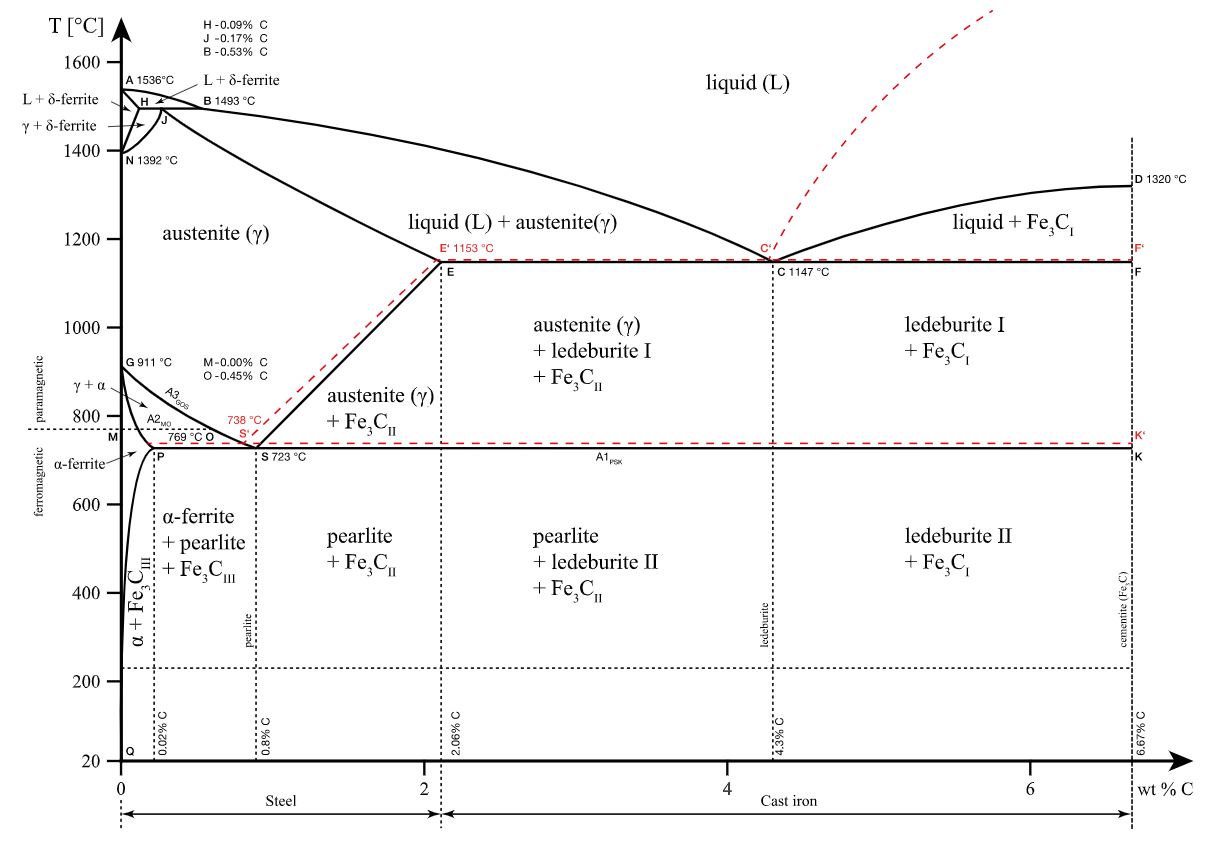
15 2 1 lattice parameter and lattice parameter expansion of pure iron.
Pure iron at room temperature has the bcc structure.
It has a hardness of approximately 80 brinell.
However iron can also be found in the fcc structure at higher temperatures.
Calculate the radius of an iron atom in this crystal.
The primary phase of low carbon or mild steel and most cast irons at room temperature is ferromagnetic α fe.
Iron ï j fp 3 crystallizes in a bcc unit cell at room temperature.
Data is presented only for the first 114 elements as well as the 118th hydrogen through flerovium and oganesson and predictions are given.
When it dissolves in iron carbon atoms occupy interstitial holes.
Predict the lattice parameter of fcc iron if it did form at room temperature assuming that atoms touch only along the most closely packed directions in both the fcc and bcc structures.
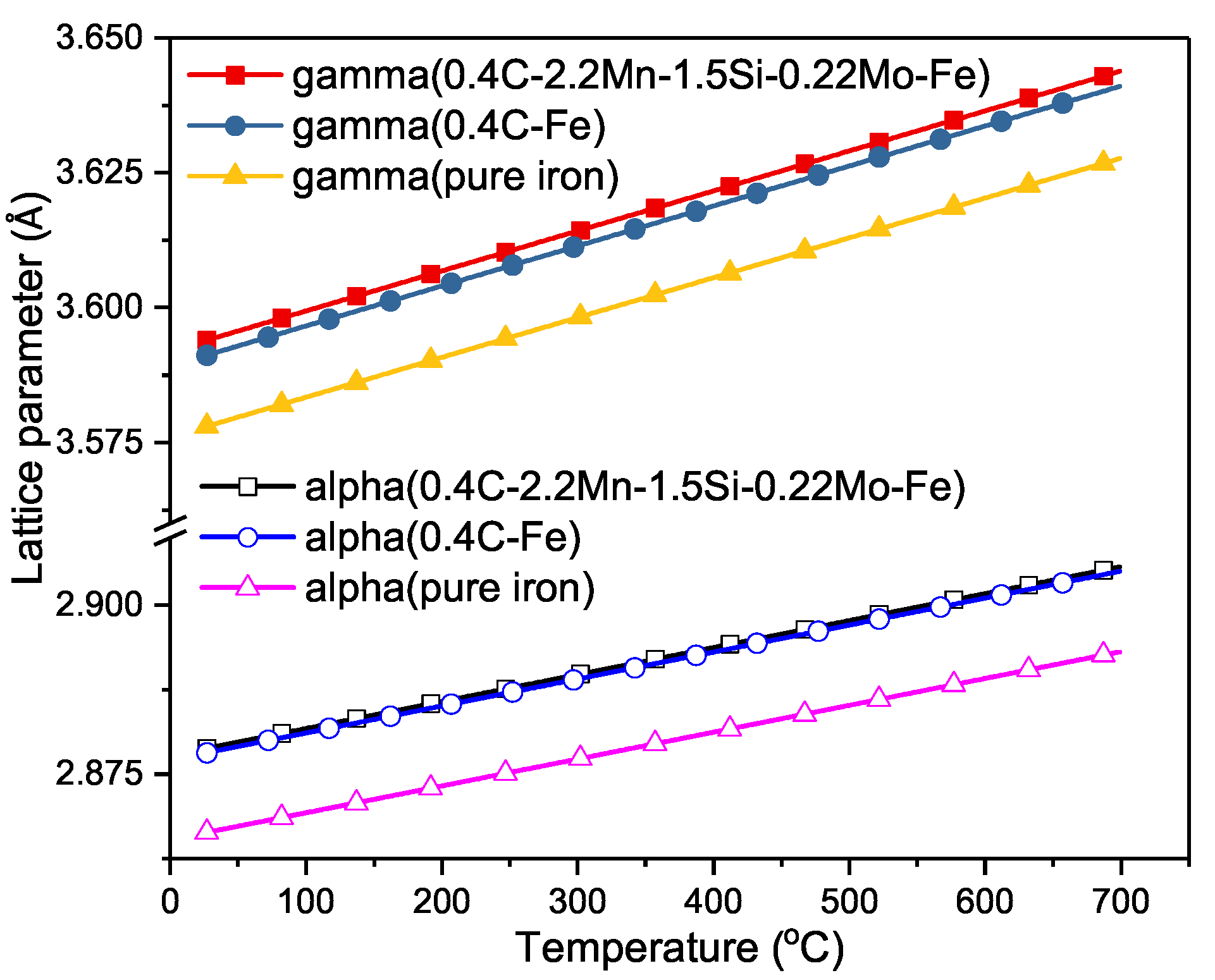
3 lattice parameters of fe c and fe n 513 3 1 3 0 7 d 2 9 2 8 t 0 from a comparison with the lattice parameter of pure f c c iron at room temperature 3573 nm.
At temperatures above 910ºc iron prefers to be fcc.
The maximum solubility is about 0 02 wt at 727 c 1 341 f and 0 001 carbon at 0 c 32 f.
15 2 2 lattice parameter of α fe ferrite with binary additions of transition metals.
The smallest repeatable three dimensional pattern is then a cube with an atom at each corner plus one in.
Section 2 it can be concluded that analogous to retained iron carbon austenite retained iron nitrogen austenite in fe n specimens overall experiences a compression.
The best known example of allotropy is exhibited by iron which at normal one atmospheric pressure is bcc at temperatures below 910 c and above 1394 c but fcc between 910 c and 1394 c as is illustrated in fig.
19 2 3 lattice parameter of γ fe austenite in literature.
In all other cases the structure given is for the element at its melting point.
Being about twice the diameter of the tetrahedral hole the carbon.